Trump FDA Commissioner Marty Makary is out

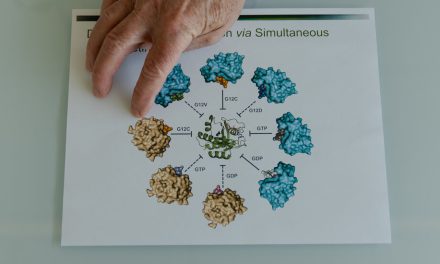
Food and Drug Administration Commissioner Marty Makary speaks during an announcement at the Department of Health and Human Services on December 18, 2025 in Washington, DC.
Alex Wong | Getty Images
Dr. Marty Makary resigned as FDA commissioner on Tuesday, ending a controversial tenure at the health agency.
In a post on Truth Social, President Donald Trump said that Kyle Diamantas, who previously served as the FDA’s top food official, would step in as acting commissioner. In earlier remarks to reporters, Trump called Makary a “wonderful man” and said, “He’s going to go on and live a good life.”
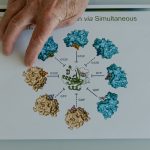
Trump shared a second post that showed an alleged resignation text from Makary. In the message, Makary noted his role in the FDA’s priority review process, which expedited approval of some drugs, among other changes at the agency.
Enlarge symbolArrows point outwards
A Truth Social post from US President Donald Trump about FDA Commissioner Marty Makary.
Donald Trump on Truth Social
A senior administration official confirmed that Makary resigned on Tuesday after days of reports that the White House was planning to fire him. Health and Human Services Secretary Robert F. Kennedy Jr. made the decision to replace Makary, the official added. The government hopes to name its candidate to replace Makary in the coming weeks, although the official warned that it was too early to name possible successors.
Makary, a surgical oncologist known for criticizing the government’s handling of the Covid pandemic, was head of the agency responsible for regulating food, drugs and medical devices for more than a year.
His tenure has been marked by internal dysfunction and turmoil in the FDA’s leadership, as well as increasing backlash from drugmakers, doctors and patient groups over regulatory decisions, including high-profile rejections of some treatments for rare diseases. At the same time, the White House reportedly grew increasingly impatient with what it saw as slow action on Trump’s key policy initiatives, such as the legalization of flavored e-cigarettes.
The influential organization Susan B. Anthony Pro-Life America had also called for Makary’s resignation over the FDA’s handling of the abortion pill mifepristone. Makary has reportedly been slowly conducting a safety review of the pill, which can be mailed to states that restrict abortion. Makary’s successor will inherit these criticisms and the sensitive politics surrounding abortion.
The senior administration official said they could not point to a single problem that led to Makary’s ouster, but said it was an accumulation of many problems. Abortion was one of those issues.
Despite the controversy surrounding recent drug rejections, the pharmaceutical industry appears to be concerned about a shakeup at the top of the FDA. The pharmaceutical industry is negotiating to reauthorize the Prescription Drug User Act Fee, which sets the fees the FDA charges drugmakers to fund its reviews.
The morale of the agency’s employees had fallen following layoffs and departures of career agency scientists, including long-time cancer commissioner Dr. Richard Pazdur, who cited Makary’s leadership qualities as the reason for his departure. Meanwhile, distrust of management among the remaining employees is said to have grown.
Among Makary’s most polarizing appointments was Vinay Prasad, who served as a key official at the agency overseeing vaccines and biotech treatments before resigning in late April. Prasad, an outspoken academic and podcaster, left the agency after criticism of the FDA grew from the biotech and pharmaceutical industries, as well as from former health officials.
For example, the FDA initially refused to review Moderna’s flu shot — a decision the biotech company said conflicted with the agency’s previous guidance and specifically came from Prasad. The FDA later changed course on the vaccine.
Prasad also faced backlash earlier this year for rejecting uniQure’s gene therapy for Huntington’s disease. The company claimed the FDA required it to perform fake brain surgery to assess whether the treatment worked. In a CNBC interview in March, Makary appeared to criticize this treatment without naming it.
In April, the FDA rejected Replimune’s melanoma drug candidate for the second time, following an initial rejection in July. The authority referred to insufficient evidence of effectiveness and criticized the design of the single-arm study.
In an interview with CNBC in May, Makary said that three independent teams had come to the same conclusion about the drug and that the FDA had not made any “corrupt sweetheart deals.”
“I don’t work for Replimune, I work for the American people and I stand with the scientists at the FDA,” Makary told CNBC’s David Faber.
In March, Sen. Ron Johnson, R-Wisc., announced an investigation into the FDA’s rejection of treatments for rare diseases.
To install a new commissioner, Trump will likely need to secure the support of Sen. Bill Cassidy, a former doctor who nearly blocked the confirmation of Health and Human Services Secretary Robert F. Kennedy Jr. Trump has endorsed a candidate seeking to unseat the Louisiana Republican, which could complicate that effort.
Choose CNBC as your preferred source on Google and never miss a moment from the most trusted name in business news.